Will blockchain combat drug counterfeiting in Africa?
Every year, about 100,000 deaths are recorded in Africa due to counterfeit drug, experts say that blockchain technology is a solution to this menace.

One out of every 10 medical products in low- and middle-income countries, which includes most of Africa, is sub-standard or fake, this leads to at least 100,000 deaths in on the continent, says the World Health Organization (WHO). Also, 42% of all fake medicines reported to the WHO between 2013 and 2017 were from Africa.
At his friend's pharmacy in Ghana, Dare Odumade witnessed the death of a patient whose kidney ruptured due to a counterfeit antimalarial medicine.
In 2018, he founded Chekkit, a Nigeria-based B2B software company to provide product authentication and distribution tracking solutions to FMCGs and Pharmaceuticals. With Chekkit's technology patients and medics are able to identify counterfeits–the company raised a $500,000 pre-seed in 2021 to improve its blockchain-powered technology.
At the time of the pre-seed, Chekkit disclosed that it has secured about seven million pharmaceutical products and protected over 200,000 consumers. The company also expanded its offering into Afghanistan.
Recently, a Nigerian healthtech company, Afrihealth launched Rigour+ for manufacturers. Rigour+ is a blockchain-backed serialisation technology address drug counterfeiting which cost the global pharmaceutical industry about $200 billion annually.
"By assigning a unique identifier to each product unit, manufacturers and importers can track their products throughout the supply chain in real-time. The app's innovative geofencing tool takes this a step further by allowing manufacturers to monitor the geographical journey of their products, ensuring that they remain within intended market boundaries," says Linda Obi, CEO of Afrihealth.
Using blockchain against counterfeit drugs
According to Obi, who is also a blockchain expert:
Blockchain technology is emerging as a revolutionary force in the global fight against drug counterfeiting. By leveraging its unique attributes of transparency, traceability, and security, it promises to significantly bolster the integrity of the pharmaceutical supply chain.
- Unparalleled Transparency: Blockchain operates as a decentralized ledger that chronicles every step of a drug's journey from the manufacturing unit to the end consumer. This ledger is simultaneously shared across multiple participants - manufacturers, wholesalers, retailers, and even patients, allowing all parties a lucid view of the drug's provenance and journey. In such a transparent system, any attempt to infiltrate the supply chain with counterfeit products is likely to be swiftly detected.
- Immutable Traceability: The core strength of blockchain is its immutable nature - once data is inscribed onto the blockchain, it is resistant to modification or deletion. This feature facilitates real-time tracking and tracing of every drug unit, enabling stakeholders to spot irregularities or deviations instantly. Any discrepancy in the drug's journey becomes a glaring indicator of potential counterfeit intrusion.
- Robust Security: The underlying strength of blockchain lies in its sophisticated cryptographic protocols. Each transaction, or 'block', is intricately connected to the preceding one, forming an unbroken chain. Any attempt to tamper with a transaction disrupts the chain's continuity, triggering an immediate system-wide alert. This level of security makes it extremely challenging for counterfeiters to manipulate the data, ensuring the drug's authenticity.
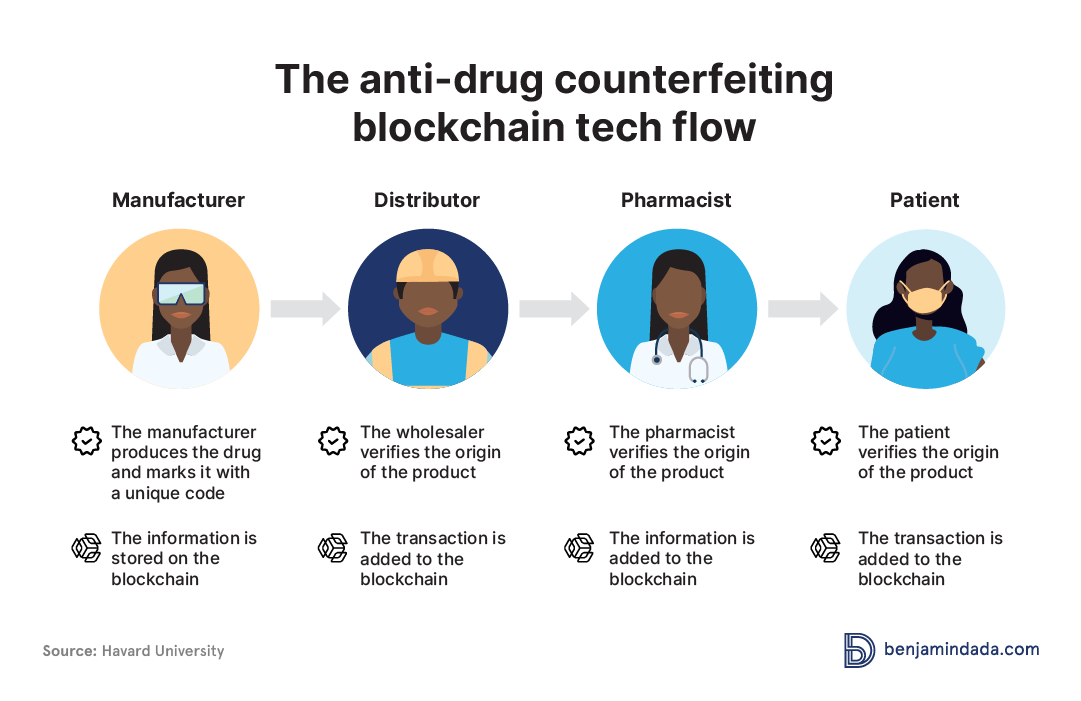
Thus, by providing an unassailable record of the pharmaceutical supply chain, blockchain technology can effectively safeguard against the infiltration of counterfeit drugs. It offers the prospect of a future where the authenticity of drugs reaching consumers is guaranteed, marking a significant stride in the battle against drug counterfeiting.
"When the promise of technological advancement meets the power of the blockchain, the result is not just progress - it is a revolution. A revolution that ensures health for all Nigerians, breaking the barriers of distance, affordability, and authenticity," Obi added.
In Nigeria, NAFDAC has a plan
Earlier this year in Nigeria, the National Agency for Food and Drug Administration and Control (NAFDAC) said it will confront the menace of substandard and falsified medicines in the country with the implementation of a pharmaceutical traceability strategy.
According to Obi, NAFDAC's push for serialisation and traceability, driven by the urgency to protect public health and the economy from the scourge of counterfeit drugs, could be a major catalyst in the adoption of advanced digital technologies in the pharmaceutical supply chain.
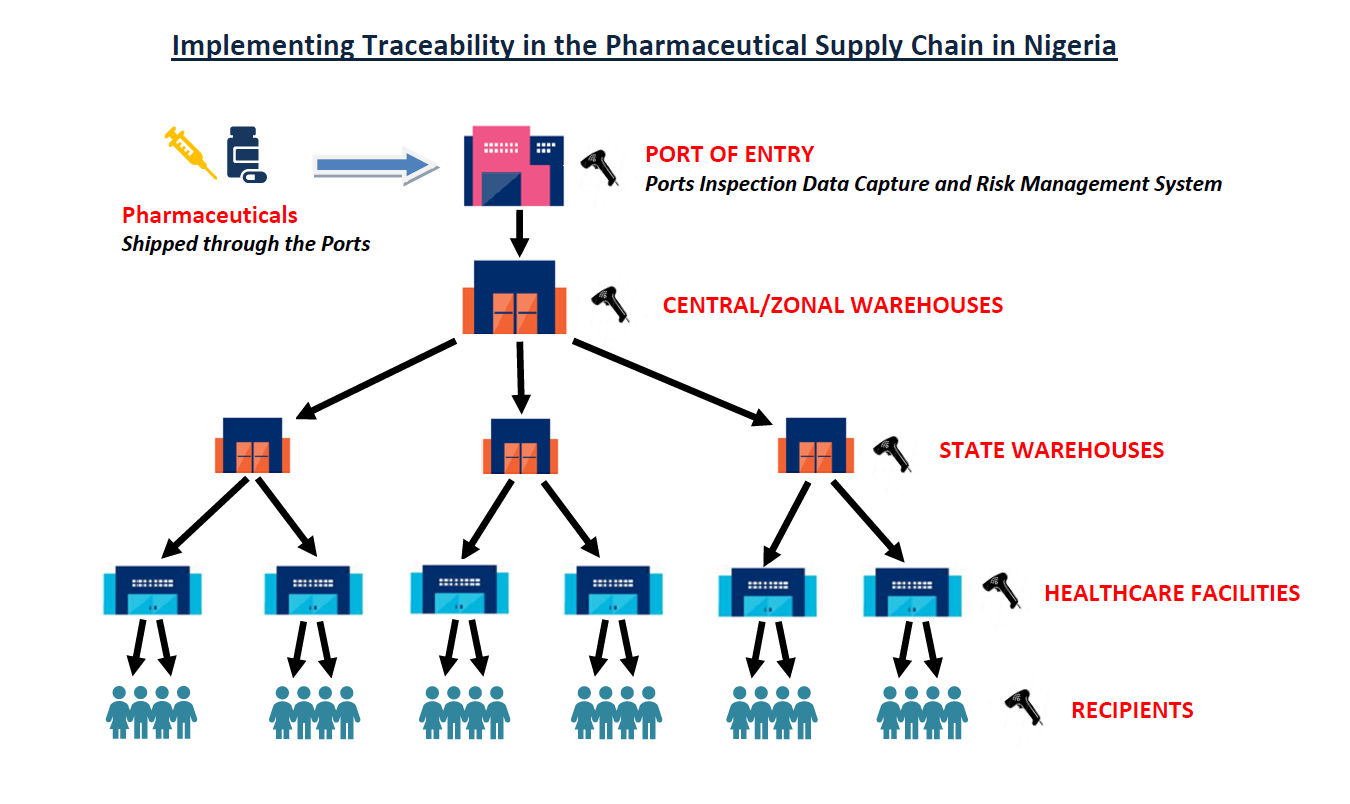
"By mandating end-to-end serialization and traceability, NAFDAC is addressing this dual crisis head-on. Serialization allows every unit of a product to be uniquely identified, making it easier to spot counterfeits," she added. "Traceability ensures that the journey of a product through the supply chain can be tracked and verified, making product diversion more difficult. The potential for substantial fines or even imprisonment for failure to comply will further deter counterfeiters."
This regulation will further drive the adoption of digital technologies like Chekkit and Rigour+. Linda agrees: "NAFDAC's push for serialization and traceability, driven by the urgency to protect public health and the economy from the scourge of counterfeit drugs, could be a major catalyst in the adoption of advanced digital technologies in the pharmaceutical supply chain."







Comments ()